Postdoc and PhD positions at the Soft Matter Group
There are no open positions at the Soft Matter group at the moment
Student projects at the Soft Matter Group

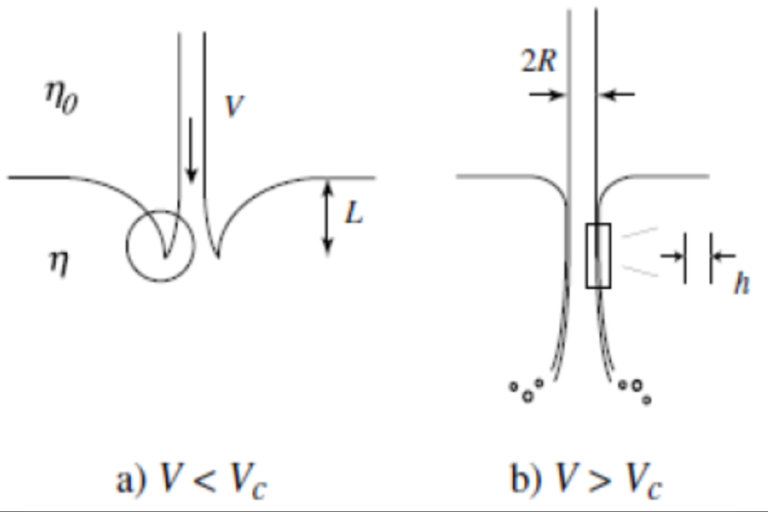
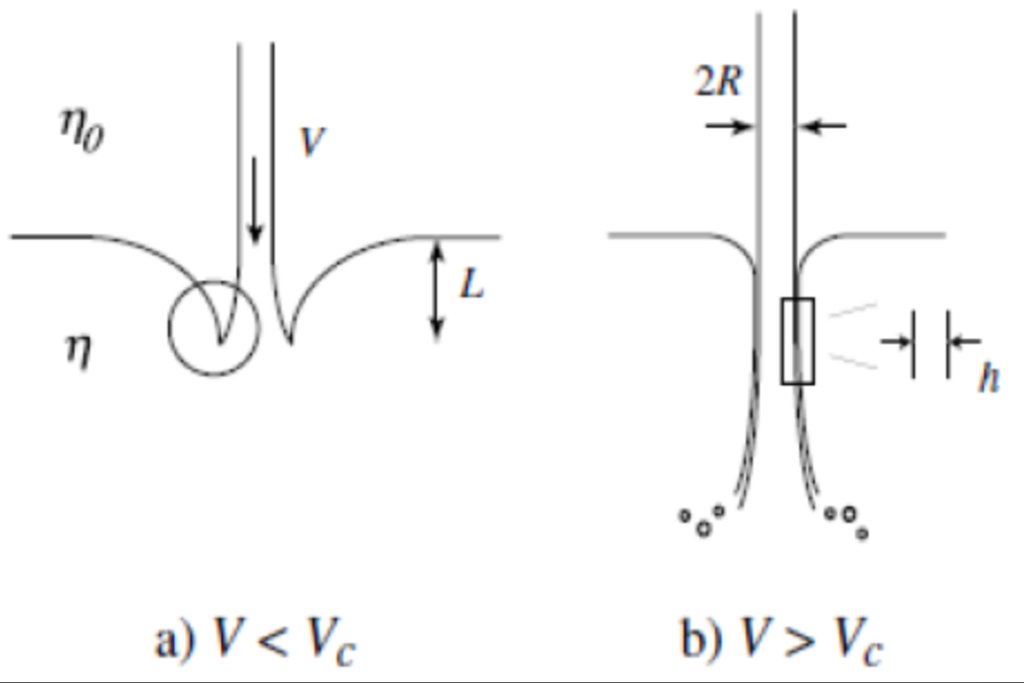
Microdrop formation with innovative nozzles
Master or Bachelor project with Cees van Rijn
Show Details 


Water at negative pressure
Master or Bachelor project with Daniel Bonn and Sander Woutersen
Show Details 

Does a growing crystal exert pressure in a confined area?
Master project with Noushine Shahidzadeh and Daniel Bonn
Show Details 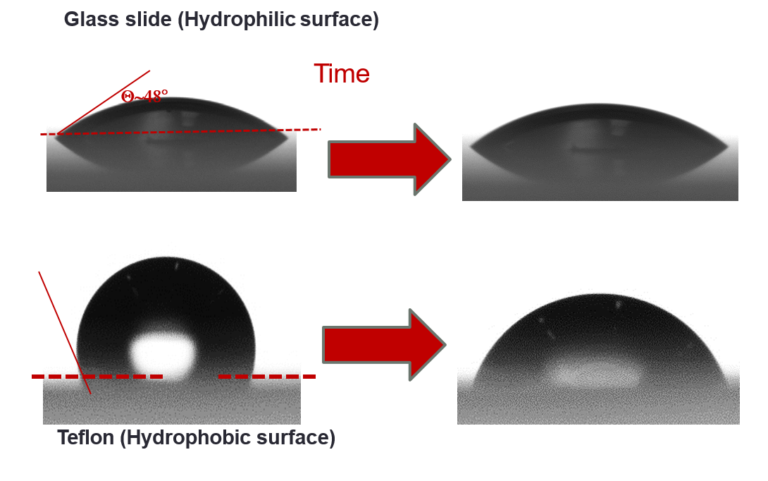
Impact of salts and surfactants on droplet spreading
Bachelor project with Noushine Shahidzadeh and Daniel Bonn
Show Details 
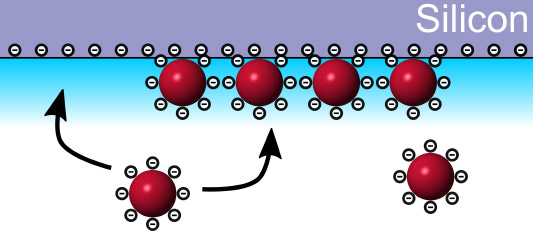
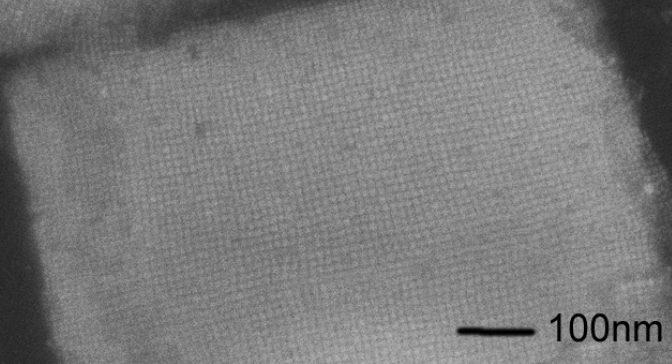
Assembly of MoS2 Quantum Dots into Thin Films
Master or Bachelor project with Peter Schall
Show Details 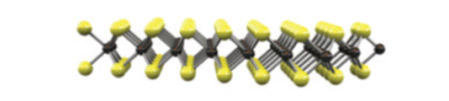
Optical Properties of Mechanically Exfoliated Bi2S3
Master or Bachelor project with Peter Schall
Show Details