 Back to projects
Back to projects
Colloids as big atoms
- Supervisor: prof. Peter Schall
- Research Team: Piet Swinkels
- Goal: Explore assembly of atom using colloids, which are visible with microscopy
- Info: P. Swinkels, P. Schall
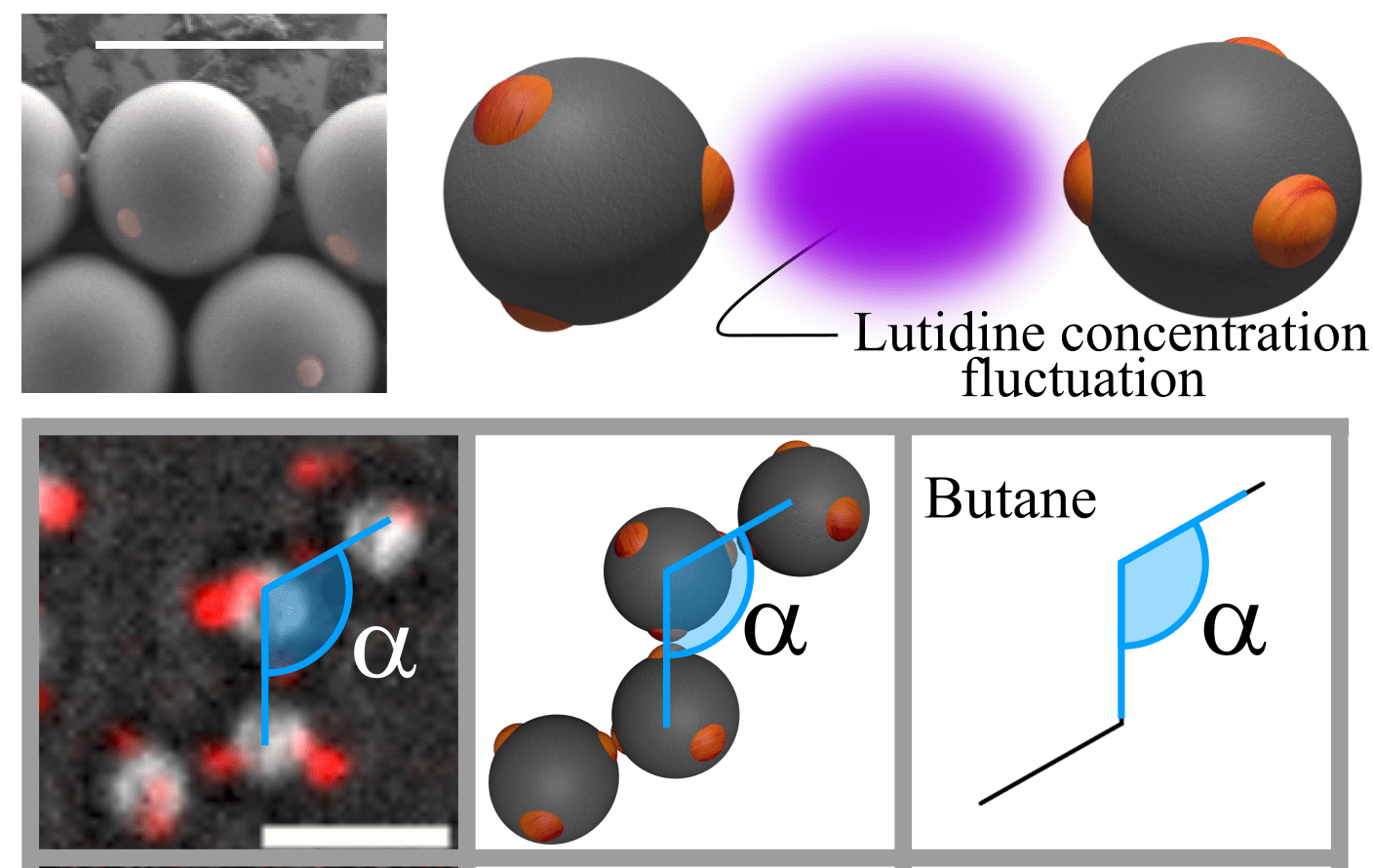
Colloidal particles offer fascinating insight into the statistical mechanics and assembly behaviour of atoms. The particles, about a micrometer in size, have thermal energy and attractive/repulsive interactions similar to atoms, making them form phases very similar to their atomic counterpart. Yet, due to their larger size, they can be easily observed by optical microscopy. Besides being atomic models, these particles serve as building blocks for the assembly of new micro- and nanoscale materials that are used, e.g. in photonics and optoelectronics. One of the things we can do is bind “patchy” colloidal particles into colloidal molecules using critical Casimir interactions. The attractive critical Casimir force arises only between the patches, from the confinement of critical solvent fluctuations between them.Colloids with directed interactions assemble into colloidal molecules, with structures mimicking that of atomic molecules. The figure shows colloidal molecules assembled from di- and tetrapatch colloidal particles. Optical microscopy images are shown on the left, and their molecular analogues on the right. These particles form molecular compounds known from organic chemistry, and because of their size can be directly observed at the particle scale by microscopy. Furthermore, their three-dimensional structure and dynamics can conveniently be observed. As the colloidal molecule is in a highly thermally excited state, where quantum mechanical states become quasi continuous, it corresponds to the classical limit of its atomic counterpart, and as such presents a good model of the actual molecule. The behaviour of the colloidal molecule matches the behaviour of the atomic case surprisingly well. For instance, in colloidal cyclopentane, we observe the familiar puckering and pseudorotation of the ring. By introducing a surface wich can bind patches, we can even catalyse ring-opening reactions.
